|
The half-life is 301,200 years and can be found naturally in trace quantities. Chlorine-36 decays by - decay into 36 Ar 98.1 of the time or 36 S by electron capture 1.9 of the time. 
It is formed when cosmic radiation interacts with atmospheric chlorine. This is the reason why H is always a terminal atom and never a central atom. Chlorine-36 is a radioactive isotope containing 19 neutrons. Hydrogen only needs to form one bond to complete a duet of electrons. Atom (Group number)īecause hydrogen only needs two electrons to fill its valence shell, it follows the duet rule. Table showing 4 different atoms, each of their number of bonds, and each of their number of lone pairs. In each case, the sum of the number of bonds and the number of lone pairs is 4, which is equivalent to eight (octet) electrons. The number of electrons required to obtain an octet determines the number of covalent bonds an atom can form. The gas is toxic.Relative atomic mass: 35.446, 35. It is a greenish-yellow, disagreeable gas with irritating odour. Oxygen and other atoms in group 16 obtain an octet by forming two covalent bonds: The origin of the name comes from the Greek word chloros meaning pale green. To obtain an octet, these atoms form three covalent bonds, as in NH 3 (ammonia). Group 15 elements such as nitrogen have five valence electrons in the atomic Lewis symbol: one lone pair and three unpaired electrons. 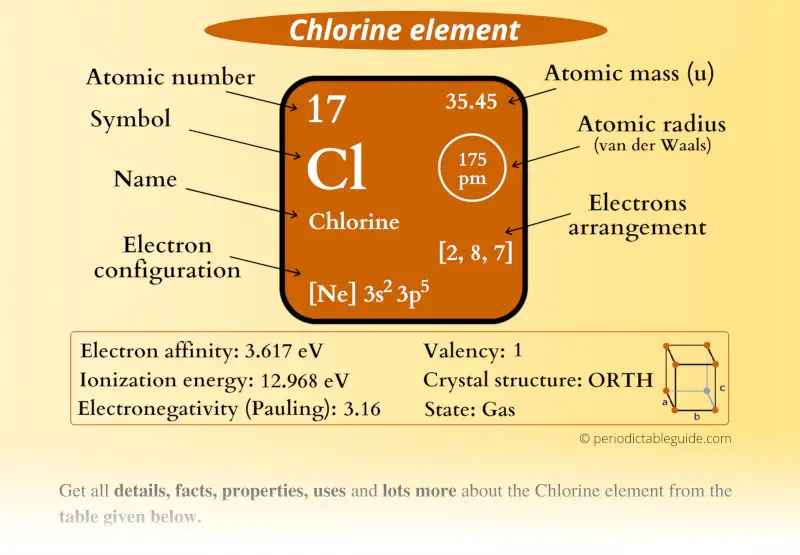
The transition elements and inner transition elements also do not follow the octet rule since they have d and f electrons involved in their valence shells. Because hydrogen only needs two electrons to fill its valence shell, it is an exception to the octet rule and only needs to form one bond. These four electrons can be gained by forming four covalent bonds, as illustrated here for carbon in CCl 4 (carbon tetrachloride) and silicon in SiH 4 (silane). For example, each atom of a group 14 element has four electrons in its outermost shell and therefore requires four more electrons to reach an octet. The number of bonds that an atom can form can often be predicted from the number of electrons needed to reach an octet (eight valence electrons) this is especially true of the nonmetals of the second period of the periodic table (C, N, O, and F). From left to right: water molecule, ammonia molecule, and methane molecule They have created a complete suite of short videos on every element on the periodic table of elements.\): 3D molecule examples. The video below is from the University of Nottingham's periodic videos project. IsotopesĬhlorine has two isotopes found in nature: Symbol Chlorine gas is highly toxic and was used as a chemical weapon in the First World War. In the past, chlorine was used to make chloroform and carbon tetrachloride (cleaning solvent). It's an oxidising agent and an excellent substitution element. Ĭhlorine is an element with many applications in chemistry. Other chlorine compounds are used in the manufacturing process for bullet-proof vests, parachutes, cockpit canopies, and so on. Chlorine is also a component of polyvinyl chloride (PVC) plastic which is used for things like window frames, car interiors, electrical wiring insulation, and water pipes. It's used in almost every product that requires disinfecting at some stage of its production. It's used in water, ranging from drinking water to swimming pools. Ĭhlorine is primarily used as a disinfectant. Chlorine liquid at 7.4 bars of pressure in an ampoule and embedded in glass.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
